
our mission:
cure desmoid tumors
The Desmoid Tumor Research Foundation (DTRF) empowers and unites everyone committed to finding answers for the desmoid tumor community.
We aggressively fund desmoid tumor research, accelerate the development of improved therapies, drive collaborations among clinicians and researchers worldwide, and support patients and doctors through education. Our goal is to find a cure for each and every patient diagnosed with this rare disease.
Every year we gain extraordinary momentum in our progress. Our research and collaboration efforts have led to FDA approval of the first-ever medical therapy to treat desmoid tumors, as well as other emerging therapies, new clinical trials, and important discoveries about the disease.
As we find more answers, we also work tirelessly to provide patients and caregivers with the resources, information, and opportunities needed to be informed advocates for their care.
We accomplish our mission by focusing on the following strategic areas
Research Funding
The DTRF will stop at nothing to find more treatments and ultimately, a cure for desmoid tumors. We’re the world’s leading foundation dedicated to desmoid tumor research, advocacy, awareness, and support.
We are guided by our Advisory Boards, comprised of top multidisciplinary experts from prestigious institutions around the world. Their counsel informs which research the DTRF funds to advance desmoid tumor science and drive the development of new treatment options.
DTRF-funded research has already made significant advancements in our understanding of the disease.
Education & Support
Patients are not fighting this disease alone. Our website provides the education and support needed to navigate their journey and make important decisions about their health. Our events, including our annual DTRF Together We Will Weekend, connect the patient community with experts in the field and empower them with knowledge about the latest research and treatment options.
Our website also serves as an information hub for clinicians, researchers, and others in the medical community, supporting them in their goals of clinical improvements and advances in research.
Collaboration & Connection
One of our most impactful priorities is facilitating collaboration and connection. As a key thought leader and advocate in the desmoid tumor landscape, we are in the unique position of seeing things from the point of view of the patient, clinician, and researcher. This allows us to identify and advance initiatives that benefit our entire community.
Throughout the year, we provide opportunities for our patients to connect with each other at our virtual and in-person events. Clinical trials and Patient-Powered Research projects connect patients directly to research.
Through the DTRF Coordination Core researchers are connected to findings and data. Our Virtual Tumor Board brings clinicians together around individual cases, while the DTRF Int’l Desmoid Tumor Research Workshop fosters collaboration on research priorities and education about treatments.
Our efforts have brought about breakthrough results in record time for a rare disease. Listen to DTRF’s Scientific Director Dr. Mrinal Gounder and co-founder Marlene Portnoy recount the story of how this collaboration connected patients to a clinical study named one of the top 10 oncology breakthroughs of 2018 by the American Society of Clinical Oncology (ASCO).
These are just some examples of the impacts of collaboration facilitated by the DTRF between patients, clinicians, researchers and industry partners.
Our Purpose
The truth is that rare diseases do not get the same attention or funding as more common diseases. It takes a grassroots team of patient advocates like DTRF to bring awareness to the cause and build a network of people dedicated to finding a cure.
Dr. Ben Alman of Duke University on DTRF’s 15th Anniversary
Advocacy Group Partners
We know that our goal of a cure is within reach, and we will get there together.
We forge local and international collaborations among desmoid patient groups outside the US, and with rare disease groups that share common goals and priorities. We are proud to work alongside all of the international desmoid patient advocacy groups through our Annual Virtual Global Desmoid Tumor Roundtable, as well as rare disease groups such as the National Organization for Rare Disorders (NORD), My Pediatric and Adult Rare Tumor (MyPART) Network, SARC, the Sarcoma Alliance, and others committed to the identification, treatment, and cure of rare disorders through programs of education, advocacy, research, and patient services.
Corporate Partners
WE BELIEVE IN CONVENING key stakeholders to help accelerate desmoid tumor research and expand treatment options for the patient community.
Our corporate partners support our mission by connecting with the DTRF team on a regular to basis to better understand the needs of the patient community and learn about the advocacy efforts constantly in motion at the Foundation.

Your Donation Changes Lives
When you donate to the Desmoid Tumor Research Foundation (DTRF), you help fund research toward better treatment options and a cure for each and every desmoid tumor patient. Every donation, no matter the size, helps us continue our mission. We are so grateful for your contribution.
The DTRF is a Connecticut non-stock, nonprofit corporation, determined by the IRS to be a tax-exempt Section 501(c)(3) organization. Donations are deductible to the extent allowed by law. Federal Tax ID number: 61-1493017.
Continue Exploring

Our Strategy & Impact

How a Labor of Love Turned into a Global Effort to Find a Cure
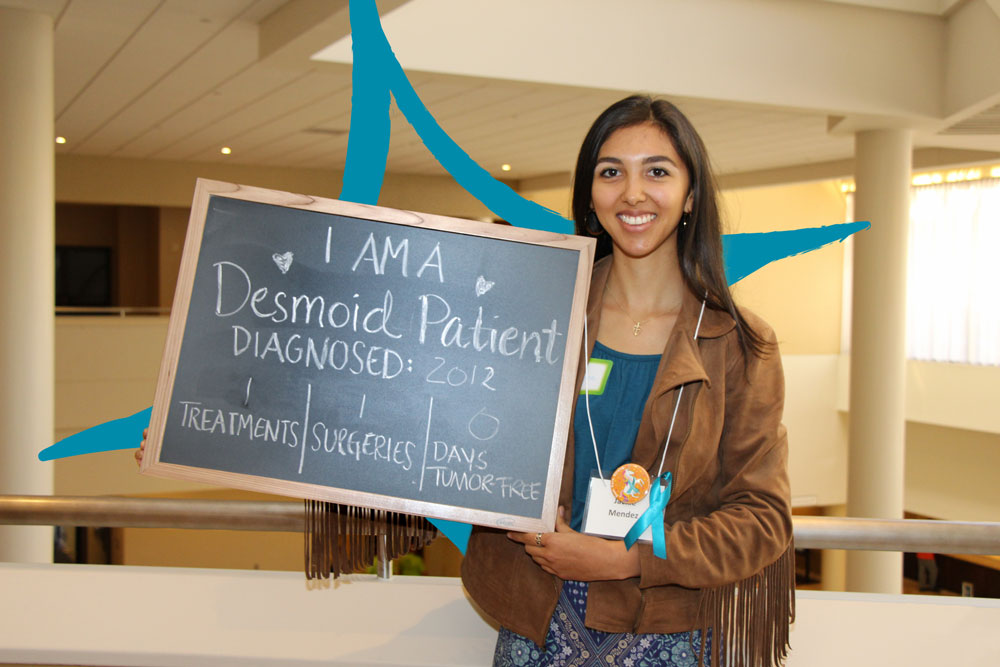
Your Donation Changes Lives
When you donate to the Desmoid Tumor Research Foundation (DTRF), you help fund research toward better treatment options and a cure for each and every desmoid tumor patient. Every donation, no matter the size, helps us continue our mission. We are so grateful for your contribution.
The DTRF is a Connecticut non-stock, nonprofit corporation, determined by the IRS to be a tax-exempt Section 501(c)(3) organization. Donations are deductible to the extent allowed by law. Federal Tax ID number: 61-1493017.





 .
. 